(Suction Activated Patent Hemostasis)


SAPH System
Full SAPH system with sheath
Syringe pulling plunger
SAPH Band
How it Works
SAPH is designed to achieve hemostasis after transradial procedures—without collapsing the artery. Here’s how it works:
1. Align
The integrated notch guides precise positioning over the sheath entry site.
2. Activate
A 20 cc syringe generates gentle negative pressure, drawing the arteriotomy toward the soft pad.
3. Secure
A one-way valve maintains the vacuum seal; no manual titration or incremental deflation required.
4. Release
After the prescribed dwell time, the valve is opened to equalize pressure and allow removal of the device.
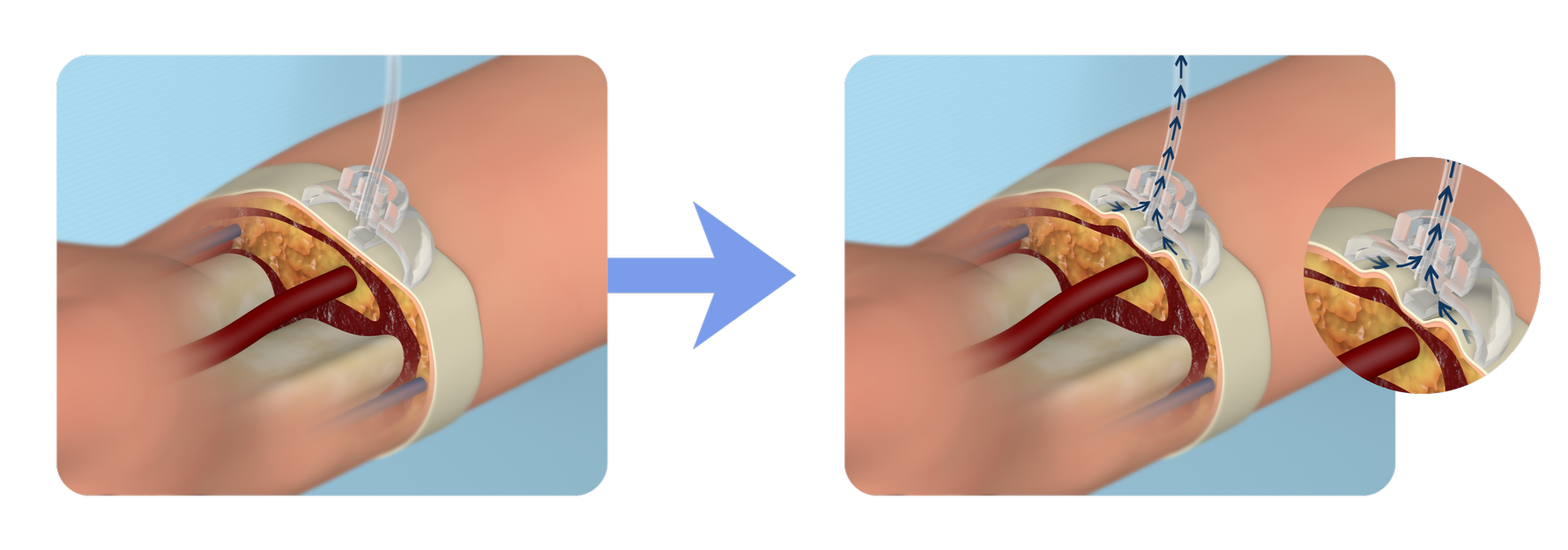
Mechanism of Action
Under controlled suction, SAPH gently lifts the skin and subcutaneous tissue, approximating the arteriotomy and tissue between the arteriotomy and device to a soft external seal. This topical apposition is designed to support hemostasis without circumferential compression to help preserve antegrade flow through the radial artery. Intrinsic blood pressure and clotting factors then contribute to natural healing.
Radial Closure is Still a Compression Workflow
Compression-based closure typically requires:
staged deflation protocols
repeated nursing checks
pulse monitoring
bleeding management
reinflation cycles
SAPH Changes the Closure Workflow
Because the mechanism does not depend on titrated compression, the closure process is designed to become:
simpler to manage
less dependent on staff expertise
more predictable for recovery teams
Potential implications include:
reduced bedside intervention
more consistent hemostasis management
improved recovery workflow
Early Clinical Experience (Data on File)
17-patient first-in-human series:
100% acute hemostasis; 0% RAO observed; no device-related complications. Early clinical experience not powered for definitive safety or effectiveness conclusions.
Prospective 50-patient study ongoing to evaluate workflow efficiency, safety, and comfort
Designed for regulatory submission under FDA 510(k) pathway (predicate classification: vascular clamp/ compression, 21 CFR 870.4450).
SAPH is not yet FDA cleared, it is for investigational use only.
Aligned with Published Best Practices
SAPH is designed in accordance with published radial-first access and patent-hemostasis protocols that emphasize:
Preservation of arterial flow
Reduction of RAO Risk Factors
Enhanced patient comfort and recovery
